|
7/5/2023 0 Comments Entropy of vaporization 
(b) Calculate the enthalpy of vaporization of methanol, assuming that rS doesnt depend on temperature and taking the boiling point of methanol to be 64.6C. (a) Calculate the entropy change for the vaporization of 1 mol methanol (use data from Table 16.1 or Appendix J). Once we get all the values of entropy change, just add them up to get the standard molar entropy of vaporization of water. The standard molar entropy of methanol vapor, CH3OH(g), is 239.8 J K1 mol-1. This was the best explanation for this problem! I just followed these steps and finally got the right answer :) What is the boiling point(C) of a liquid with entropy of vaporization 68.3 JK1mol1 and enthalpy of vaporization 35.65 kJ mol1: rounded up to two decimal. As the degree of the disorder increases in the transition, from a liquid having a small volume to a vapour having a larger volume, the entropy of vaporisation is always positive. 
Once we get all the values of entropy change, just add them up to get the standard molar entropy of vaporization of water. The entropy of vaporisation is the increase in entropy upon the vaporisation of a liquid. Just plug and solve to get that entropy change The entropy of vaporization at the normal boiling point has been estimated for binary and ternary azotropic mixtures containing water, alcohol or acetic. Only this time, we use the Cpm 33.6 J⋅K−1⋅mol−1 for water vapor and the temperature change is from 100 ☌ to 59 ☌. To solve the third part, we use again the equation ∆S = Cpm ln (T2/T1). Since the Cpm is 75.3 J⋅K−1⋅mol−1 for liquid water and the temperature change is from 59.0 ☌ to 100 ☌, just plug and solve to get the entropy changeĪs for the second part, we already are given the entropy of vaporization of water at 100 ☌ which is 109.0 J⋅K−1⋅mol−1. To solve the first part, we should use the equation ∆S = Cpm ln (T2/T1). Entropy of vaporization is an increase in entropy upon vaporization of a liquid.To solve this problem, we need to know three steps that involve finding the standard molar entropy of vaporization: the entropy change heating water from 59.0 ☌ to 100 ☌, the entropy of vaporization of water at 100.0 ☌, and the entropy change cooling water from 100 ☌ to 59 ☌. Note: Enthalpy of vaporization is the amount of energy that must be added to the liquid substance, to transform a quantity of that substance into gas. This maximum can be attained only in a completely reversible process. 1 atm.In thermodynamics, the Gibbs free energy is a thermodynamic potential that can be used to calculate the maximum of irreversible work that may be performed by a thermodynamic system at a constant temperature and pressure.The Gibbs free energy is the maximum amount of non-expansion work that can be extracted from a thermodynamic closed system. Std entropy change of fusion, fus S o 31 J/(mol·K) Std enthalpy change of vaporization, vap H o 42.3 ± 0. The change in free energy occurs when a compound is formed from its elements in their most thermodynamically stable state at standard state conditions i.e. Latent Heat of Vaporization are saturated properties, just enter One. ∴ Boiling point of the liquid at one atmospheric pressure is 400k. Pressure: Density: Volume: Quality: Internal Energy: Enthalpy: Entropy: Isochoric.  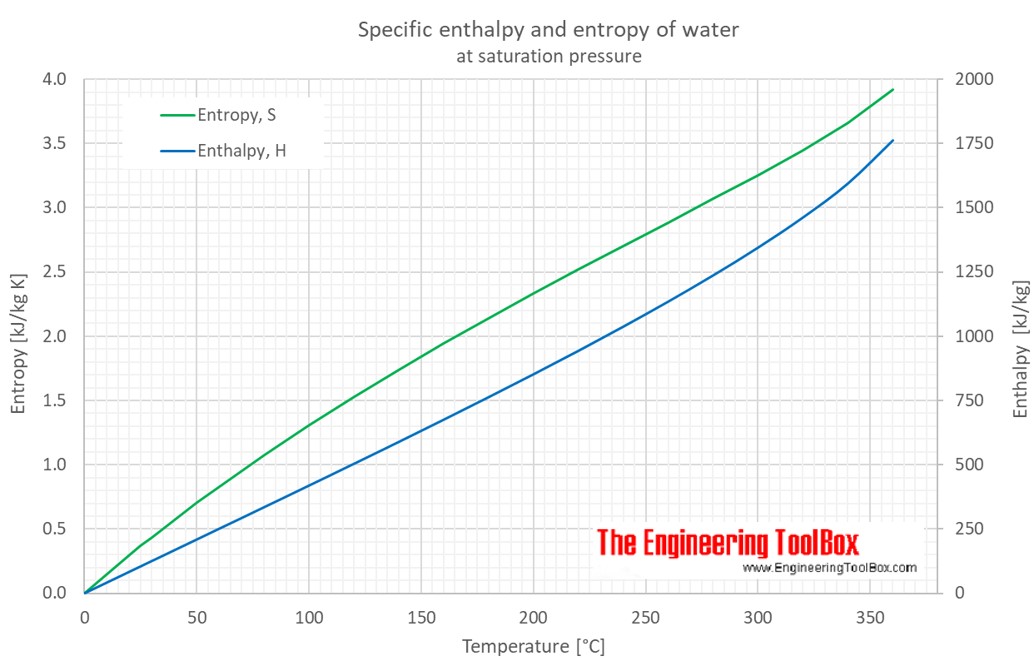
$\Delta H$ = enthalpy of vaporization = 30000 $Jmo$ The entropy change for the system and its surroundings is identical in magnitude but opposite in direction, resulting in a zero entropy universe. It is given that Enthalpy of vaporization is 30 Kilojoules per mole. Entropies Equation of state Internal energies Isobars Isochores Isotherms Joule - Thomson inversion Latent heats of vaporization Melting. We know that at equilibrium, Gibbs energy change is zero. See Table 11.7 for heats of vaporization. vapor is in equilibrium at one atmospheric pressure. Calculate the change in entropy that occurs in the system when 1.00 mole of diethyl ether (C4H10O) condenses from. Hint: the fact that at boiling point of the liquid, liquid
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
